Seven company cards
Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Key themes
Across 7 large biotech exits, probably the most important theme is to have Sanofi buy you if you’re a bit earlier. Similar to the 2000s/2010s theme of startups partnering with Celgene.
The average time from founding date to exit was 13.8 years, with a median of 11 years. And an average valuation of $5.6B at acquisition with the 2 largest outcomes, the closest to clinical approval.
Kymab showed the value, and difficulty, of pioneering a new MoA. And taking a variant view on immunology in the backdrop of a boom.
Ablynx had a technology monopoly on nanobodies and might have undersold itself depending on how its candidates do in the clinic this decade. ChemoCentryx has similarities here pioneering chemokine medicines. As well as Affinivax, building a better version of existing vaccines.
Biohaven filled a clear gap for biopharma in neuroscience and provides a template for inventors & business model innovation in biotech
Acceleron had a clear biological hypothesis that drove their clinical activities/updates over 15 years. With Global Blood Therapeutics following a similar, maybe clearer, path.
All together, biotech companies get started with platforms & acquired for products. But the bar for assets get a lot higher where the market/technology has less interest from biopharma (i.e. neuro/infectious disease).
Kymab was acquired by Sanofi for >$1B in 2021. It is a case study of developing drugs with high conviction despite patent challenges, competition in the clinic, & trailblazing a new MoA. Founded in 2010 out of Cambridge, UK, Kymab is also an example for every British biotech.
The platform is centered around a transgenic mouse model from the Allan Bradley Lab at the Sanger Institute. Kymab was able to generate a mAb for OX40L (an emerging cancer target) among other immunoregulatory targets https://www.kymab.com/media/uploads/files/Lee_et_al._2014-1.pdf
This technology led to patent challenges from Regeneron, settled in 2020 https://www.kymab.com/media/uploads/files/2020-06-24_Kymab_UK_Supreme_Court_Press_ReleaseFINAL.pdf
Despite operating in this backdrop, Kymab was able to drive their lead to the clinic. In 2020, they reported phase 2a data in severe atopic dermatitis meeting both primary endpoints.
In an IO boom where companies were focusing on increasing/decreasing T-cell subpopulations. Kymab focused on balancing them. Driving their clinical success, & partnership discussions leading to Sanofi’s (new CEO wanted to do deals) acquisition https://www.sanofi.com/en/media-room/press-releases/2021/2021-01-11-07-30-00-2155914
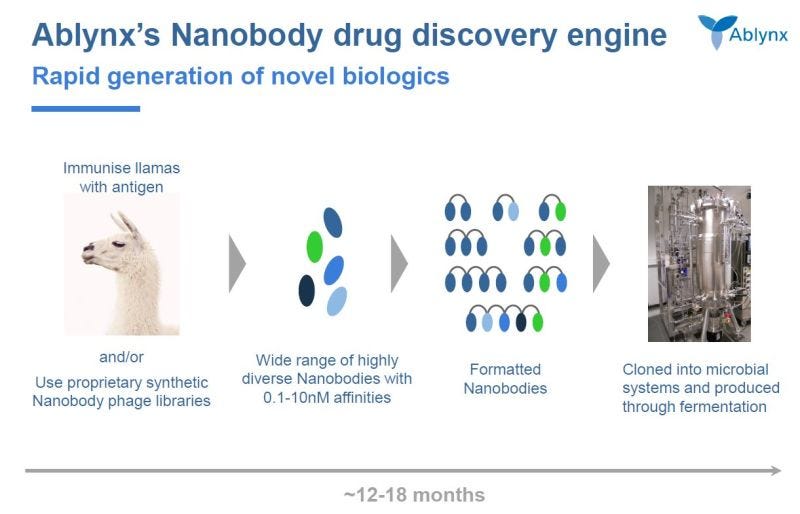
Ablynx pioneered nanobodies, got acquired for €3.9B by Sanofi in 2018, and might have massively undersold itself. Founded in 2001 based on research out of Brussels, the company can trace its origins back to a Moroccan camel in the 1990s to a drug approval in 2019.
Nanobodies = single-domain antibody fragments, combining the advantages of conventional antibodies with some features of small molecules. They were initially found in camels and llamas & are significantly smaller than human antibodies https://originofimpact.sites.vib.be/en/nanobodies
Ablynx built a near technology monopoly with 100s of patents & even owning the nanobody trademark. Their lead asset, caplacizumab, an anti-von Willebrand factor nanobody, treating aTTP was approved in 2019 targeting $100Ms in peak sales. Setting up a decade of new nanobody trials.
In retrospective, depending on the success of caplacizumab and other drug candidates, Ablynx might have sold itself too early. Learn more about Ablynx and their platform here: https://www.sanofi.com/en/science-and-innovation/research-and-development/technology-platforms/nanobody-technology-platform
Biohaven was acquired by Pfizer in 2022 for $11.6B. Founded in 2013, led by Vlad Coric & based on his lab’s work, Biohaven was started to fill the gap in neurology drug development biopharma left. Ultimately leading to the approval of Nurtec to treat migraines.
Biohaven built a pipeline around 3 main mechanisms: calcitonin gene-related peptide (CGRP) receptor antagonists, glutamate modulators, & myeloperoxidase inhibitors. The lead (i.e. Nurtec) blocks the CGRP receptor to get at the root cause of migraines & avoid vasoconstriction.
With over 1B people suffering from migraines, Pfizer acquired Nurtec. After the acquisition, Biohaven was able to keep all non-CGRP development candidates along with >$200M in cash to continue on its goal to develop best-in-class medicines in neurology.
Biohaven is a template for all inventors to follow and an inspiration for all neuroscience drug development companies. Biohaven is hiring here: https://www.biohaven.com/careers/
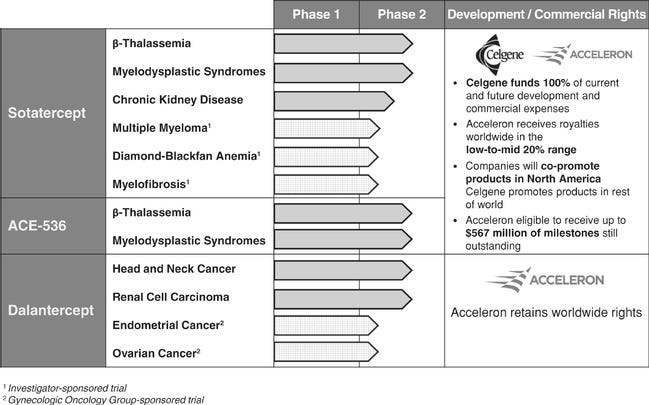
Acceleron was acquired by Merck for $11.5B in 2021. Initially working on bone loss & anemia, Acceleron’s key decision was to focus on pulmonary arterial hypertension (PAH) with sotatercept. Generating exciting enough late-stage clinical data to compel Merck to buy them.
Founded in 2003, Acceleron started trials in 2006, built partnerships with Celgene, & went public in 2013. The company was centered around protein therapeutics to target the TGF-β superfamily of ligands/receptors. Their initial pipeline focused on cancer and blood disorders.
This work ultimately led to the approval of Reblozyl in 2020 for anemia, alongside Celgene/BMS. The key inflection point was developing sotatercept (ligand trap to rebalance BMPR2 signaling) for PAH, a rare disease leading to heart failure https://www.merck.com/news/merck-to-acquire-acceleron-pharma-inc/
Existing medicines promote dilation while sotatercept tries to reverse muscularization. With Merck reporting phase 3 data (N=323) in March 2023, reducing risk of death by ~84% compared to placebo https://www.nejm.org/doi/full/10.1056/NEJMoa2213558
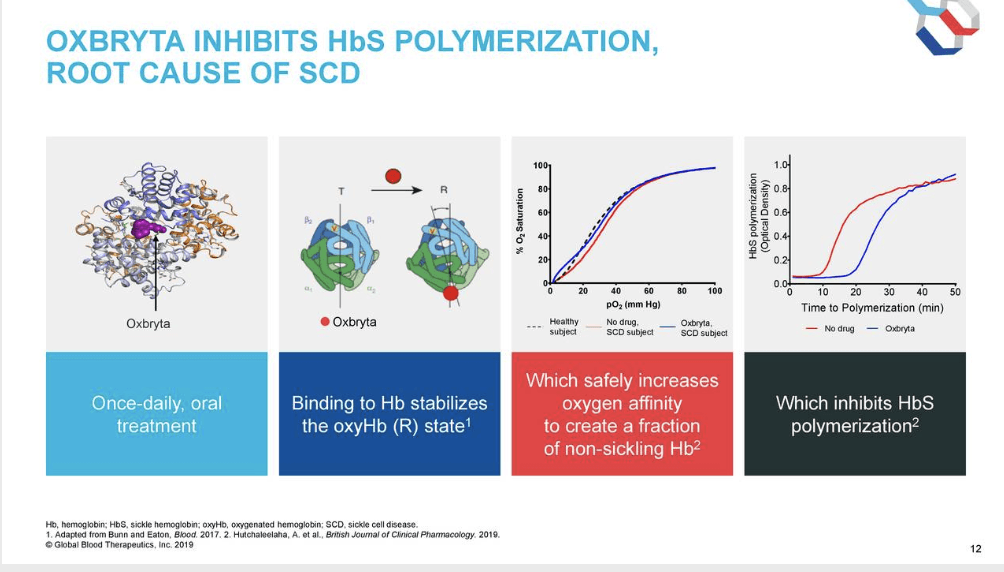
Global Blood Therapeutics was acquired by Pfizer for $5.4B in 2022. Founded in 2011, GBT used its SHAPE Platform to discover small molecules for key blood proteins. Which led to the development of Oxbryta, approved in 2019 as an oral, once-daily treatment for sickle cell disease.
SCD is caused by a single mutation in hemoglobin that alters in shape & ability to transport oxygen. The active substance in Oxbryta, voxelotor, works by improving the ability of the hemoglobin to hold on to oxygen & preventing it from forming chains.
GBT has been able to treat 1000s of patients with Oxbryta so far. With sickle cell disease affecting ~100K patients in the US and well over 15M around the world, there is still a lot of work to expand access to the medicine.
Learn more here: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-completes-acquisition-global-blood-therapeutics
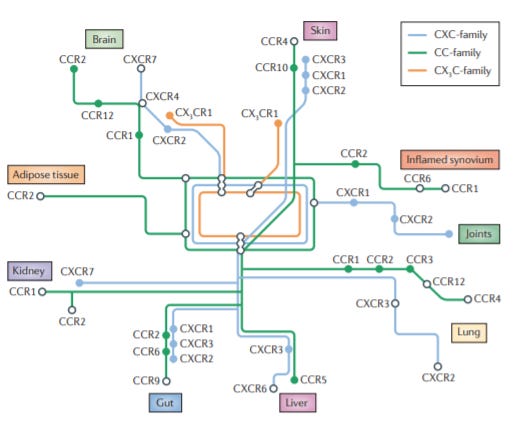
ChemoCentryx was acquired by Amgen for ~$3.8B in 2022. Founded in 1996, by Thomas Schall who discovered many of the 50 chemokines, the company was started to drug them, the trafficking control system for immune system & develop once-daily, orally-available drugs for autoimmunity.
Chemokines act as signposts to get different immune cells into certain organs. ChemoCentryx built a pipeline to target chemokines & receptors to treat autoimmunity & inflammatory diseases. This led to the development/approval of avacopan insevere active ANCA-associated vasculitis.
Most autoimmune medicines are either injectable or given through IV. By creating a more user-friendly drug, ChemoCentryx is positioned to capture large parts of the autoimmunity market. The company has candidates in development for hidradenitis suppurativa, C3G, & more.
Thomas Schall is a template for all founders & inventors to follow and ChemoCentryx is beginning to transform the lives of patients https://www.amgen.com/newsroom/press-releases/2022/08/amgen-to-acquire-chemocentryx-for-$4-billion-in-cash
Affinivax was acquired by GSK in 2022 for $2.1B. Founded in 2014 out of Boston Children’s Hospital, Affinivax develops vaccines without adjuvants. The company was acquired mainly for its 24-valent pneumococcal vaccine in late-stage trials to go heads up against Pfizer/Prevnar.
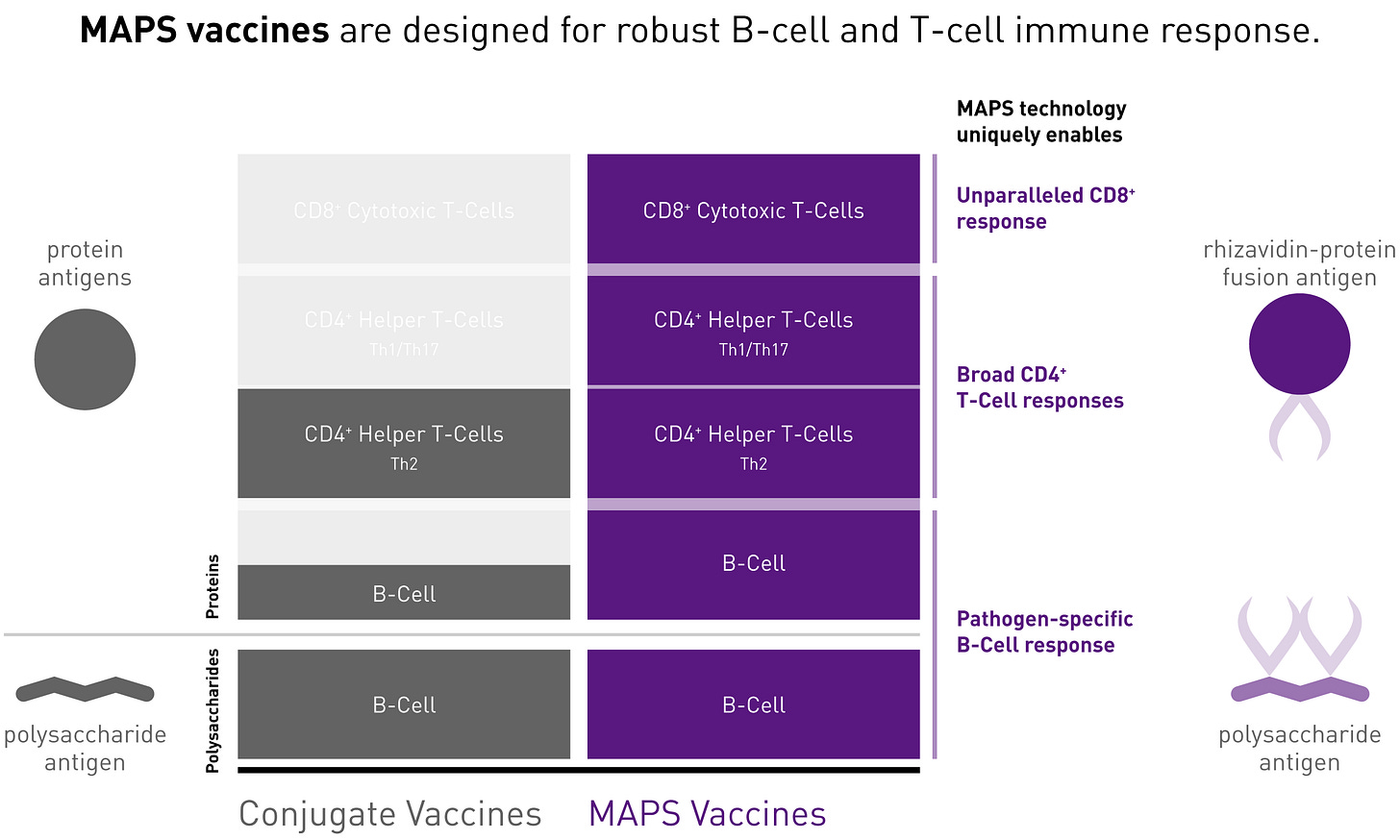
The company is built around its MAPS platform that uses non-covalent chemistry between biotin & rhizavidin to conjugate/present both polysaccharide & protein antigens to the host immune system to induce a B- and T-cell immune response. MAPS = Multiple Antigen Presenting System.
MAPS enables higher valency than conventional conjugation technologies. Affinivax’s focus is pneumococcal disease, the main cause of acute bacterial infections, leading to >150K annual hospitalizations in the US. Pfizer’s Prevnar has >$5B/year in PD, the bar is pretty high here.
Affinivax shows the value in vaccine development still exists. The initial focus is on preventing pneumococcal disease with a purview on other infectious diseases especially those acquired in a hospital https://www.gsk.com/en-gb/media/press-releases/gsk-completes-acquisition-of-affinivax-inc/